Prostate Cancer
Cytovac focused initially on patients with prostate cancer, since these patients have significant treatment needs not covered by existing treatments. The patients enrolled in these studies (CV001 and CV002) are late-stage prostate cancer (Castrate Resistant Prostate Cancer (CRPC)) patients. Both patients with and without metastasis were included.
The phase I testing of ALECSAT showed the treatment to be well tolerated, safe and applicable for prostate cancer patients. Several positive indications for clinical effect were observed as assessed by a transient decrease or stabilization in prostate specific antigen (PSA) levels, but due to the low number of patients enrolled, no statistically valid efficacy conclusions could be made.
Based on the clinical phase I data, ALECSAT treatments of prostate cancer should be followed up in a clinical phase II trial. Cytovac seeks partners to perform such a trial.

- CV001 (PHASE IA) “FIRST DOSE IN MAN – SAFETY OF A SINGLE ALECSAT DOSE IN LATE STAGE PROSTATE CANCER”
- CV002 (PHASE IB) “SAFETY OF REPEATED DOSING OF ALECSAT IN LATE STAGE PROSTATE CANCER``
- PROSTATE CANCER – CONCLUSION
In 2009, Cytovac initiated the first clinical trial in prostate cancer. This was a prospective, open-label, single center, phase I trial of 12 weeks duration, to investigate the safety and tolerability of a single dose of ALECSAT in 13 patients with late-stage castrate-resistant prostate cancer (CRPC). The trial was completed in 2010. ALECSAT treatment was found to be well tolerated and no serious side effects that could be associated to the treatment were seen.
For several patients, positive indications of disease improvement were observed as assessed by a transient decrease or stabilisation in prostate specific antigen (PSA) levels.
The ALECSAT productions, logistics and handling of treatments in the hospital were successful.
In 2011, Cytovac initiated a second larger prospective, open-label, single centre, phase Ib prostate cancer trial of 25 weeks duration. The trial was completed in October 2012. Whereas the first trial (CV001), was a single-dose trial, the 21 patients included in this second phase Ib trial, were treated with 3 doses of ALECSAT. The primary objective was to investigate the safety and tolerability of ALECSAT in patients with late-stage castrate-resistant prostate cancer (CRPC). The secondary objective was to assess indications of a positive therapeutic or palliative effect of ALECSAT.
Repeated ALECSAT treatments were seen to be well tolerated and no serious side effects that could be associated to the treatments were seen. For several patients, indications of possible disease improvement were observed as assessed by decreases or stabilization in prostate specific antigen or by improvements in bone metastases observed by skeletal scintigraphy (reduced number and/or growth of metastases).
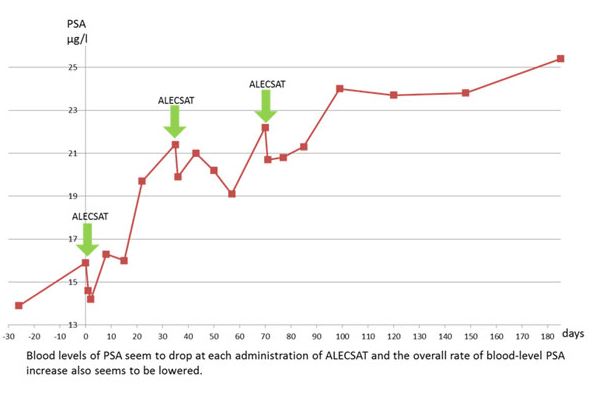
Based on the 2 completed clinical trials in prostate cancer it can be concluded:
- The ALECSAT production, logistics and handling of treatments in the hospital were successful
- Treatment with ALECSAT was generally well tolerated and no serious side effects associated with ALECSAT treatment were seen,
- Although the patients were at a late stage of their disease, several patients showed indications of disease improvement :
– Stabilization or decrease in PSA levels (rise in PSA indicates disease progression)
– Reduction in size and/or number of bone metastasis.
These results are encouraging, however, as only few patients were treated in each trial these preliminary results should be further corroborated.

