Brain Cancer – Clinical Development Status
Glioblastoma multiforme (GBM) is one of the most malignant types of central nervous system tumors and remains largely incurable. With current existing treatments, the prognosis is poor and the overall survival is short. Thus, there is a large unmet medical need for new and better treatments that can improve the outcome for this group of patients. Based on the encouraging results from the trials in prostate cancer, three clinical trials have been conducted in patients with GBM brain cancer.
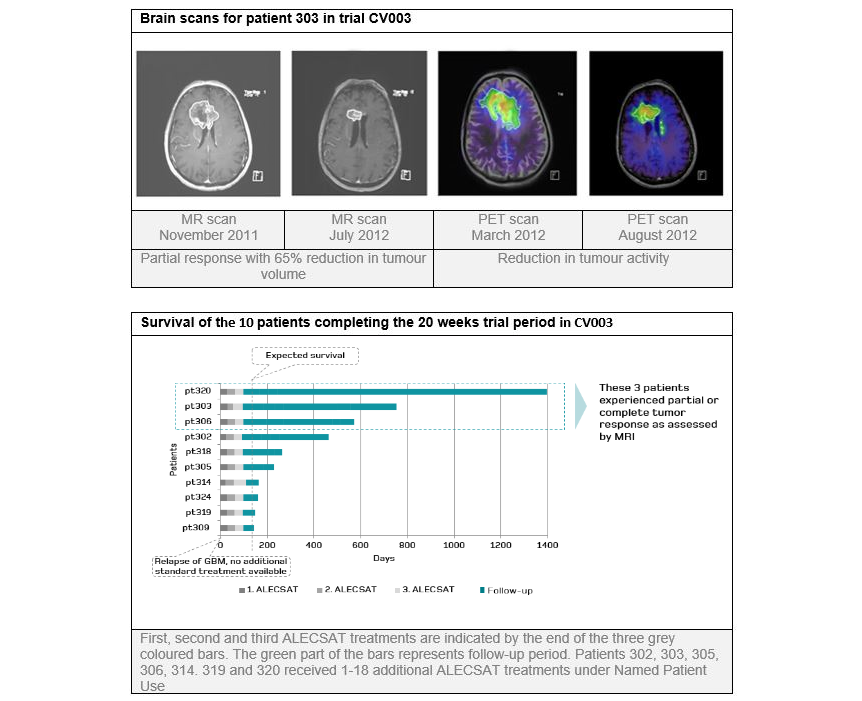
- CV003 (PHASE IB) “SAFETY OF REPEATED DOSING OF ALECSAT IN THE TREATMENT OF LATE STAGE GBM”.
- CV005 (CLINICAL PHASE II): ”EFFICACY AND SAFETY OF REPEATED DOSING OF ALECSAT IN TREATMENT OF LATE STAGE GBM, COMPARED TO BEVACIZUMAB/IRINOTECAN”.
- CV006 (PHASE II): “EFFICACY AND SAFETY OF ALECSAT AS ADD-ON TO RADIOTHERAPY+TEMOZOLAMIDE (STUPP REGIMEN) IN NEWLY DIAGNOSED GBM COMPARED TO STUPP REGIMEN ALONE”.
In 2011, Cytovac initiated the first trial, CV003, in stage IV patients with Glioblastoma multiforme (GBM). The trial was completed in 2013. The trial was a prospective, open-label, single center, phase Ib trial of 20 weeks duration, to assess the tolerability and safety of ALECSAT treatment. 25 patients with relapsed GBM received currently best available standard treatments. The patients were to receive 3 doses of ALECSAT. However, due to the very rapid progression of this cancer disease, not all patients received the intended 3 ALECSAT treatments.
Repeated ALECSAT treatments were well tolerated and no serious side effects could be associated to the treatments.
Furthermore, among the 10 patients who received all 3 ALECSAT treatments according to protocol, several experienced reductions or stabilization in tumor size during the trial. Under approved Named Patient Use, 7 patients received 1–18 additional ALECSAT injections beyond the 20 weeks trial period. Three of these patients experienced prolonged survival with remarkable reductions in tumor size and improvement in quality of life. For the 10 patients who received 3 or more ALECSAT treatments, the average survival time was 25,6 months.
The clinical benefits observed in CV003 are supported by MRI imaging showing homing of labelled ALECSAT cells to the brain tumor site, 24 hours after administration of the labelled cells. In addition, assessment of tumor biopsies by immunohistochemistry from patients undergoing reoperation during the trial showed accumulation of CD8+ cells (cytotoxic T cells / killer cells) compared to assessment before the ALECSAT treatment.
In conclusion, the data from CV003 found ALECSAT to be overall safe and well tolerated with indications of prolonged survival for some of the 10 patients receiving 3 or more ALECSAT treatments. The observation that some patients experienced considerable reduction in brain tumor size with ALECSAT treatment encouraged additional studies.
The subsequent phase II studies CV005 and CV006 did overall not support the data from the CV003 trial. A new small trial with selected subgroups of patients suffering GBM is under investigation, aiming to identify markers predicting a positive outcome of the ALECSAT treatment.
Reference to inserted: Kirkin AF, Dzhandzhugazyan KN, Guldberg P, et al. Adoptive cancer immunotherapy using DNA-demethylated T helper cells as antigen-presenting cells. Nat Commun.,9(1):785. 2018
Following the promising results from trial CV003, a second larger comparative trial in GBM was initiated in 2014. This trial was an open-labelled, randomised, multicentre phase II trial of 62 weeks duration in patients with relapsed GBM having received the Danish recognised first-line treatments. The primary objective was to compare progression-free survival (PFS) between patients treated with ALECSAT immunotherapy and the Danish standard second line therapy with bevacizumab/Irinotecan. After 25 of the planned 175 patients had been enrolled (15 patients in the ALECSAT arm and 10 patients in the bevacizumab/Irinotecan control arm), the trial was terminated in 2015 by Cytovac. It was evaluated that the study would not reach the intended primary endpoint and advice from the European Medicines Agency (EMA) suggested, that several adjustments to the trial design were warranted. No safety concerns emerged from the trial.
Following scientific advice from EMA, this trial is designed as an open-label, randomised, multicentre, phase II trial to investigate the effect of ALECSAT treatment as an add-on radiotherapy and temozolomide (standard of care; the Stupp regimen) compared to radiotherapy and temozolomide alone in patients with newly diagnosed GBM. The trial was initiated in 2016.
The trial is ongoing at five clinical centres in Sweden; recruitment has completed.

